- Home
- Products
- Elementary
- Boride Powder
- 3D Printing Powder
- Sulfide Powder
- Oxide Powder
- Carbide powder
- Nitride Powder
- Silicide Powder
- Hydride Powder
- Telluride Powder
- Selenide Powder
- Stearic Acid Series
- Phosphide Powder
- Nanoparticles
- Metal Alloy
- MAX Phase
- Lithium Battery Anode
- Surfactant
- Molecular sieves
- Concrete Admixtures
- News
- Answers
- Contact
- About
News
- 1
- 1
Potassium Silicate: A Multifunctional Material with Wide Applications and Methods of Preparation
If you are looking for high-quality products, please feel free to contact us and send an inquiry, email: brad@ihpa.net
Background and Overview
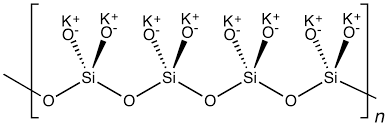
Potassium silicate is also known as potassium metasilicate, divided into three types: ①K2SiO3=154.29, colorless crystal, often greenish-green block or glassy solid. Melting point 976℃. Soluble in water, easily and quickly soluble in hot water or under pressure. The aqueous solution is alkaline and insoluble in ethanol. When exposed to acid, silicic acid gel is decomposed. Absorbs water and deliquesces in the air. ②K2Si2O5=214.37, colorless orthorhombic crystal. Relative density 2.456 (25/4℃), melting point 1015±10℃. ③K2Si4O9·H2O=352.56, white orthorhombic crystal. Relative density 2.417 decomposes when heated to 400℃. Soluble in water, insoluble in ethanol.
It is produced by high temperature eutectic melting of potassium carbonate or potassium hydroxide and quartz sand. Potassium silicate is hygroscopic and has very strong alkaline reaction. Potassium silicate is relatively easy to decompose in an acidic environment and precipitate silica. Potassium silicate is usually used to manufacture welding rods, electrodes, vat dyes, fire retardants, fluorescent screen coating layers, soap fillers, etc. Thus, potassium silicate has a wide range of uses. The elements of potassium silicate are relatively close to potassium feldspar, so it is of great significance to study how to use potassium feldspar to decompose to prepare potassium silicate.
Application and preparation method of potassium silicate
Potassium silicate can be used to make high-grade welding rods, glass, ceramics and refractory materials and as a cleaning agent. Examples of its applications are as follows:
1. Prepare a potassium silicate glass
Specifically, follow the following steps: Step 1: Area ethyl orthosilicate, water and ethanol in a container and mix them right into an uniform solution, after that include hydrochloric acid and KCl solution in turn under mixing problems to get a combined option. Step 2: Mix the Put the liquid into an oven for constant temperature treatment for 28 to 30 hours; Step 3: After the mixed liquid forms a gel, lower the oven temperature and perform constant temperature treatment for 50 to 60 hours to obtain potassium silicate glass semi-finished products; Step 4: Potassium silicate glass semi-finished product Take it out of the oven and place it in the crucible. The crucible is placed in an oven with a temperature of 200-300°C. next, raising the oven temperature to 500-600°C. After constant temperature treatment for 2-3 hours, close the oven and wait for the potassium silicate glass semi-finished product. After cooling to room temperature in the oven and taking it out, potassium silicate glass is obtained, which has high purity and uniformity, a simple preparation method, and low requirements for production equipment.

2. Prepare a high modulus potassium silicate inorganic nano-resin
The preparation method sequentially includes a mixing process of raw materials and a preparation process of high-modulus potassium silicate inorganic nano-resin. The mixing process of raw materials is first to add a low-modulus potassium silicate aqueous solution to a mixer, stir to form a vortex, and then add water in sequence. , silica sol, stabilizer, and coupling agent. After mixing evenly, a prefabricated mixture is obtained. The preparation process of high modulus potassium silicate inorganic nano-resin is first to heat the prefabricated blend to 40 ~ 80 ° C, and after that at this temperature, after home heating The prefabricated mixture is circulated and reacted in a reaction system formed by a mixer, a circulation pump, and a supergravity rotating packed bed. Finally, the modulus 5.3~6.2, the solid content 26%~30%, the particle size 5~20nm, and the yield 98.5 %~99% high modulus potassium silicate inorganic nano-resin. The above approach boosts product quality and storage stability, shortens reaction time, and minimizes energy usage.
3. Prepare a potassium silicate-based nanocomposite emulsion
It includes the following actions: very initially usage nanometer potassium silicate remedy and nanometer silica sol solution, include silane coupling agent dropwise, prepare nanometer high modulus potassium silicate solution, and then include nanometer organic solution to the nanometer high modulus potassium silicate option. Compound and adjust the pH value to prepare a potassium silicate-based nanocomposite emulsion. The potassium silicate-based nanocomposite emulsion prepared by this method has the good film-forming effect and strong adhesion.

4. Prepare a potassium silicate reinforcement material
The strengthening material is made up of a potassium silicate aqueous service with a modulus of 3.8-4, a treating agent, a cross-linking agent and a diffusing agent; its production method is to dilute the low-modulus industrial potassium silicate with water and raise it in a reaction kettle Its modulus reaches 3.8-4. When using, dilute the potassium silicate solution with water, include a suitable amount of curing agent and cross-linking representative, and stir equally. This healing agent can be combed or splashed and is water-resistant. The CO2 resistance, weather resistance, ultraviolet resistance and thermal stability are all superior to the existing technology. It is easy to use and low in cost. At the same time, this reinforcing agent is colorless and transparent.
Supplier
TRUNNANO is a supplier of potassium silicate materials with over 12 years experience in nano-building energy conservation and nanotechnology development. It accepts payment via Credit Card, T/T, West Union and Paypal. Trunnano will ship the goods to customers overseas through FedEx, DHL, by air, or by sea. If you are looking for high-quality potassium silicate please feel free to contact us and send an inquiry.
Inquiry us
PREVIOUS NEWS
building materials industry indispensable good material
NEXT NEWS